 The distribution is valid for atoms or molecules constituting ideal gases. Β = 1 / k B T, for reversible processes onlyīelow are useful results from the Maxwell–Boltzmann distribution for an ideal gas, and the implications of the Entropy quantity. General derived quantities Quantity (Common Name/s) 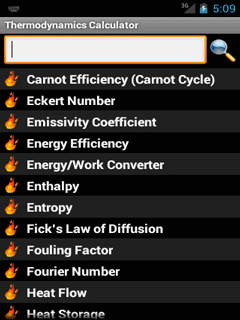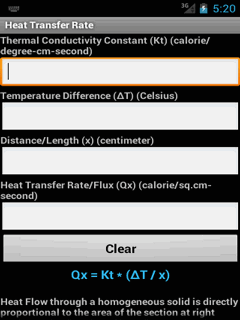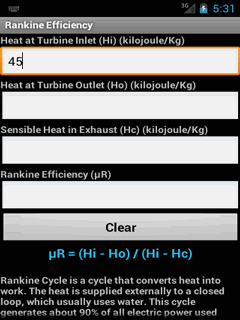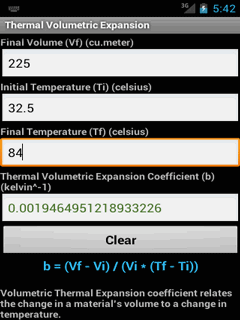 
General basic quantities Quantity (Common Name/s) for example, try this reaction: sucrose + H2O. Thermodynamics can be roughly encapsulated with these topics: Heat and Work. eQuilibrator 3.0 has been released To learn more, read the latest publication on Nucleic Acids Research. The term itself clearly suggests what is happening - 'thermo', from temperature, meaning energy, and 'dynamics', which means the change over time. Explain Hesss law and use it to compute reaction enthalpies. Many of the definitions below are also used in the thermodynamics of chemical reactions. Thermodynamics is the study of energy changes accompanying physical and chemical changes. Calculate enthalpy changes for various chemical reactions. 
The energy of a system can change due to work and other forms of energy transfer such as heat. Main articles: List of thermodynamic properties, Thermodynamic potential, Free entropy, and Defining equation (physical chemistry) Google Classroom The meaning of work in thermodynamics, and how to calculate work done by the compression or expansion of a gas Key Points: Work is the energy required to move something against a force. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
